Research
We are interested in how synapses are formed and how neural circuits function, using the nematode C. elegans as primary experimental model. The C. elegans nervous system is composed of 302 neurons, and its entire connectivity is known at the ultrastructural level. Most axons, dendrites and synaptic connections can be observed in living animals using transgenic fluorescent protein markers. In our study, we take forward genetic screening approach (see illustration) to dissect molecular genetic mechanisms controlling specific biological processes in the development and maintenance of the nervous system. We have also extended our findings from C. elegans to mice to examine mechanistic conservation and divergence in complex neuronal network.
Approaches:
- Discovering unknown genetic pathways using forward genetic screens
Discover mechanisms of synaptogenesis
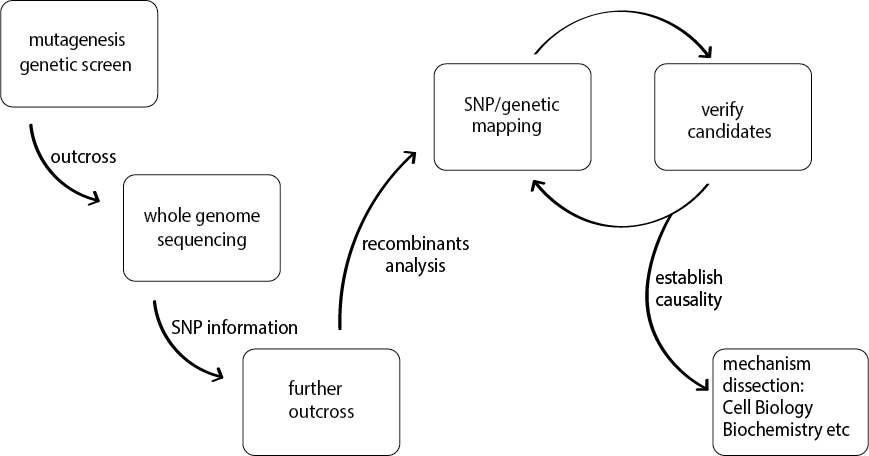
Using fluorescent markers for synaptic vesicles, we have performed numerous genetic screens. Our studies of a set of mutants led to the discovery of conserved “pioneering molecules” in synaptogenesis (see featured articles: Zhen and Jin, Nature 1999; Hallam et al., Nature Neurosci. 2001) (image below). Through hypothesis-driven, clever-designed genetic modifier screens, our subsequent studies led to in-depth mechanism discovery, for example, revealing the conserved MAPKKK DLK-1 as functionally relevant substrate of the RPM-1 E3 ubiquitin ligase (see featured articles: Zhen et al., Neuron 2000; Nakata et al., Cell 2005; Noma et al. Genetics, 2014). Our recent work has further pushing the knowledge linking synapses and unexpected themes in gene regulation (see featured articles: Chen et al., Genes & Dev. 2015). Additionally, we combine cutting-edge imagining with molecular manipulation to uncover cellular dynamics underlying neuronal circuit operation (see featured articles: Kurup et al., Current biology, 2015; Noma et al., eLife, 2017).
Imaging synapses in living animals: each fluorescent punctum represents a single presynaptic terminal
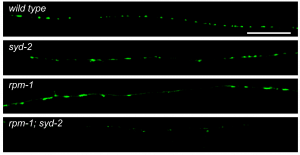
Image shows behavioral deficits for synaptogenesis mutants
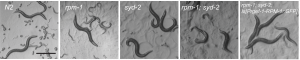
3D view of synapse
Discover axon injury and repair mechanisms
A parallel line of work in the lab has focused on addressing the ability of injured neurons to regenerate and repair damage. This is an ancient problem with close medical relevance. We developed axotomy approach to severe C. elegans axons, and defined their regenerative response. Through large scale genetic screens, we have identified multiple conserved genes affecting neuronal regenerative response (see Featured articles: Wu et al., 2007; PNAS; Chen et al., 2011 Neuron). We have also extended the novel findings to mammalian models (see featured articles: Chen et al., 2016; eLife).
An image of regrowing axon
Video of regrowing axon